Below is a sample final exam for the course. The exam is also available for download in PDF format (see below).
Question 1
Name three organelles within a cell a give a brief (one or two sentence) description of its job.
Question 2
In a few sentences, describe the difference in expression pattern between “house keeping” genes and genes whose expression is tissue specific or developmentally regulated. Give examples of four (total) tissue specific and/or developmentally regulated genes that were presented in our discussion sessions and briefly explain (two or three sentences) why each falls into that category.
Question 3
On average, what percentage of your DNA have you inherited from your paternal great-grandmother?
Question 4
Here is the nucleotide sequence for the beginning of a theoretical gene:
ATGTTTCCTAAAGAGAGGATTCGTACTGGTGCGCATCCATGAAC….
Exon 1 Intron 1 Exon 2
There are two exons and one intron shown (the intron is underlined).
What is the amino acid sequence of the protein that corresponds to this nucleotide sequence?
What is the new amino acid sequence if the two Ts at position 4 and 5 are deleted?
Question 5
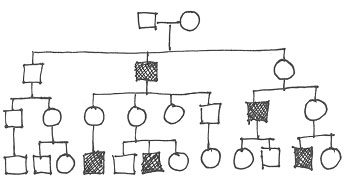
Here is a pedigree from a family in which sensorineural deafness is present in some members. Squares are males, circles are females, dark symbols are deaf family members, white symbols are normal hearing family members.
What is the mode of inheritance of deafness in this family?
Your colleague has provided you with a breeder pair of mice that each carry a heterozygous knockout of the gene “Dfns1”. In her construct, Dfns1 has been replaced by LacZ.
Here are the two alleles:
-—————-|——————-•|••• lacZ-gene ••••——-|—————|————
BamHI EcoRI BamHI EcoRI
| 2.5kb | 3kb | 2.5kb |
A B
Your colleague also gave you two short DNA fragments (A and B; shown in the drawings above) that can be used as radioactive probes to detect wild type and mutated genomic DNA.
6 weeks later, your breeder mice produced a litter of 8 animals and you prepared genomic DNA from each mouse.
6 A) What method do you use for genotyping (i.e. which animals are wild type and mutant) the animals?
6 B) What genotypes do you expect? What is the expected frequency according to Mendelian rules? (If all possible genotypes are viable)
6 C) Outline how you would be able to tell the difference between the possible genotypes? Use a drawing of a possible result of a genotyping experiment with DNA from the 8 pups and the two parents.
Question 6
Your colleague has provided you with a breeder pair of mice that each carry a heterozygous knockout of the gene “Dfns1”. In her construct, Dfns1 has been replaced by LacZ.
Here are the two alleles:
Wild type Dfns1:
Dfns1 gene
-—————-|——————-*******************|****——-|—————|————
BamHI EcoRI BamHI EcoRI
| 6kb | 1.5kb | 2.5kb |
A B
Mutated (knock-out) allele:
-—————-|——————-•|••• lacZ-gene ••••——-|—————|————
BamHI EcoRI BamHI EcoRI
| 2.5kb | 3kb | 2.5kb |
A B
Your colleague also gave you two short DNA fragments (A and B; shown in the drawings above) that can be used as radioactive probes to detect wild type and mutated genomic DNA.
6 weeks later, your breeder mice produced a litter of 8 animals and you prepared genomic DNA from each mouse.
6 A) What method do you use for genotyping (i.e. which animals are wild type and mutant) the animals?
6 B) What genotypes do you expect? What is the expected frequency according to Mendelian rules? (If all possible genotypes are viable)
6 C) Outline how you would be able to tell the difference between the possible genotypes? Use a drawing of a possible result of a genotyping experiment with DNA from the 8 pups and the two parents.
Question 7
15 years ago, your Uncle Bill was a senior graduate student in the lab of a famous researcher studying hair cell regeneration in sparrows. This was a new field at the time, and no one believed that hair cells could regrow after damage. Running a series of 2D gels*, Uncle Bill discovered a protein of about 44 kDa expressed in the basilar papilla that increases in concentration beginning 1 day after noise damage. The increased protein gradually levels off, then declines after about two weeks.
Uncle Bill named this protein BPP for basilar papilla protein. Unfortunately, Uncle Bill’s mentor was charged with scientific misconduct and tax evasion, and the lab was shut down. Uncle Bill quit graduate school and went on to become rich after opening a chain of pet food stores. Before leaving the university, Uncle Bill painstakingly purified a significant quantity of the BPP protein and still has it in his –80 degree freezer at home. He never did find out what it BPP was, but he has offered his vials of preserved BPP to you, his favorite niece/nephew.
List and explain a series of experiments that you, a modern molecular biologist, would perform on this interesting protein to try to figure out what it is and what it does. Explain why you chose to do these particular experiments and what you would learn from them. You can assume that your experiments all work, and later experiments can build upon the results of your early experiments. (They have to be real types of experiments that we’ve discussed during the course - please don’t make up wild and fantastic new protocols.) You don’t have to go into excruciating experimental details, but provide us with enough information so that we know you know what you’re talking about – the explanation for 2D-gels below is an example of about the level of detail you need.
*2D-gels are gels where you take protein extracts from a tissue or cells, and run out the extract on the gel twice – once vertically to separate based on size, and once horizontally to separate based on charge. You can do this, for example, with protein extracts made from the same tissue but at different time points, or before and after treatment, to see if there are any differences. It also serves to adequately separate proteins from each other, so you can then purify the segregated proteins from the gel, if you wish.
[We expect you to describe in detail (see above) 4 experiments of the series (that may contain more experiments)]











