This page presents selected slides and notes used during the lecture portion of the class. This includes sessions on the communications aspect of the class: oral presentations and writing reports.
Orientation
Orientation lecture by Dr. Natalie Kuldell (PDF)
Module 1: Genome Engineering
Instructors: Prof. Drew Endy, Dr. Natalie Kuldell, and Dr. Agi Stachowiak
Lecturer: Prof. Drew Endy
TA: Laure-Anne Ventouras
In this experiment, we will consider the genome of a virus, namely the bacteriophage M13. M13 is a self-assembling nano-machine with a compact genome that has been optimized by evolution to commandeer its bacterial host. Approximately 1000 new viruses are generated from a single infection event. Imagine harnessing this production. What could we build and what natural processes could we better understand? One approach we’ll take is to modify the existing genome in a subtle but useful way, namely by adding a useful sequence-tag that modifies the bacteriophage coat. We’ll examine how this modification affects the coat protein’s expression and overall phage production. Another approach we’ll take is to start from scratch, undertaking a full throttle redesign of the bacteriophage genome. We’ll employ a commercial DNA synthesis company to compile the redesigned genomic program and then we’ll see if it encoded infective M13 and if the genome of the bacterial host affects bacteriophage production. Through these investigations we’ll ask: is nature’s M13 genome “perfect” or can we do better?
Photo of M13-coated coli removed due to copyright restrictions.
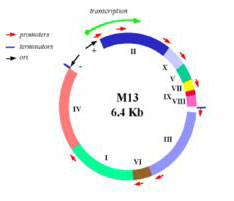
Map of M13 genome from M. Blaber. (Images courtesy of Dr. Michael Blaber. Used with permission.)
| SES # | MODULE DAY | TOPICS |
|---|---|---|
| 2 | 1 | Start-up genome engineering (PDF) |
| 3 | 2 | Agarose gel electrophoresis (PDF) |
| 4 | 3 | DNA ligation and bacterial transformation (PDF) |
| 5 | 4 | Examine candidate clones (PDF) |
| 6 | 5 |
M13.1 (PDF) Lecture on oral presentations (PDF) |
| 7 | 6 | Western analysis (PDF) |
| 8 | 7 |
Probe western (PDF - 1.1 MB) Lecture on environmental health and safety |
| 9 | 8 | Oral presentations |
Module 2: Expression Engineering
Instructors: Dr. Natalie Kuldell and Dr. Agi Stachowiak
Lecturer: Dr. Natalie Kuldell
TA: Alice Lo
In this experiment, we will consider unintended and unpredicted effects of an experimental perturbation. Our goal is a precise one, namely to silence gene expression of a measurable gene, luciferase, using RNA interference (RNAi). Each group will begin by designing a short interfering RNA (siRNA) against luciferase, but as we’ll see, siRNAs can vary in efficacy and specificity. After transfecting a mammalian cell line with the siRNA you’ve designed and a reporter plasmid, we will evaluate the silencing using a luciferase assay and microarray technology. The first assay evaluates the efficacy of the siRNA in silencing. The second assay gives genome-wide expression data to reveal the specificity of your siRNA for the gene you’ve targeted. Through this combined approach, we’ll assess the balance of targeted and off-target effects.

Child jumping through sprinkler, with sunlight. Photo by Natalie Kuldell.
| SES # | MODULE DAY | TOPICS |
|---|---|---|
| 10 | 1 | siRNA design and introduction to cell culture (PDF - 1.0 MB) |
| 11 | 2 | Transfection (PDF - 1.1 MB) |
| 12 | 3 | Luciferase assays and RNA prep (PDF) |
| 13 | 4 |
Lecture on written reports and scientific writing (PDF) |
| 14 | 5 | cDNA synthesis and microarray (PDF - 1.0 MB) |
| 15 | 6 | Microarray data analysis |
| 16 | 7 | High throughput technologies (PDF) |
| 17 | 8 | Oral presentations |
Module 3: Biomaterials Engineering
Lecturer: Prof. Angela Belcher
Content for this module is not available in OCW, because it is based on recent research that is being prepared for formal publication.










