Topics
|
|
Lecture Video
Radicals, expanded octets, and more, in this lecture about Lewis structures.
Lecture Notes
Clicker Questions
Lecture 11 Clicker Questions (PDF)
Textbook Reading
| TOPICS | 5th EDITION | 4th EDITION |
|---|---|---|
| Radicals and Biradicals | Section 2.9 | Section 2.9 |
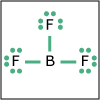
| Expanded Valence Shells | Section 2.10 | Section 2.10 |
| The Unusual Structures of Some Group 13 / III Compounds | Section 2.11 | Section 2.11 |
| Correcting the Covalent Model: Electronegativity | Section 2.12 | Section 2.12 |










