Topics
|
|
Lecture Video
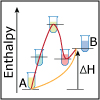
Thermodynamics is key to understanding the reactivity of molecules and compounds. In this first of three lectures on thermodynamics, viewers are introduced to ∆H, and asked to consider how much heat it will take to break one type of molecular bond versus another. Viewers are also asked whether a particular chemical reaction will release heat or absorb heat.
Lecture Notes
Clicker Questions
Lecture 15 Clicker Questions (PDF)
Textbook Reading
| TOPICS | 5th EDITION | 4th EDITION |
|---|---|---|
| The Enthalpy of Chemical Change | Sections 7.13, 7.15, 7.16, 7.17, 7.18, and 7.20 | Sections 6.13, 6.15, 6.16, 6.17, 6.18, and 6.20 |
Problems and Solutions
Solutions for Lecture 15 (PDF)
Acknowledgement
Excerpt of “Photosynthesis” by They Might Be Giants, courtesy of Idlewild / Disney Sound, used with permission. From Here Comes Science, 2009










